Ozone (O3) is an unstable gas that has a very short half-life. It reacts, then quickly disappears. The word “ozone” is derived from the Greek word for “smell” and forms naturally in the upper atmosphere where it is a vital gas that protects us from harmful UV radiation. We know it as the ozone layer.
What is Ozone?
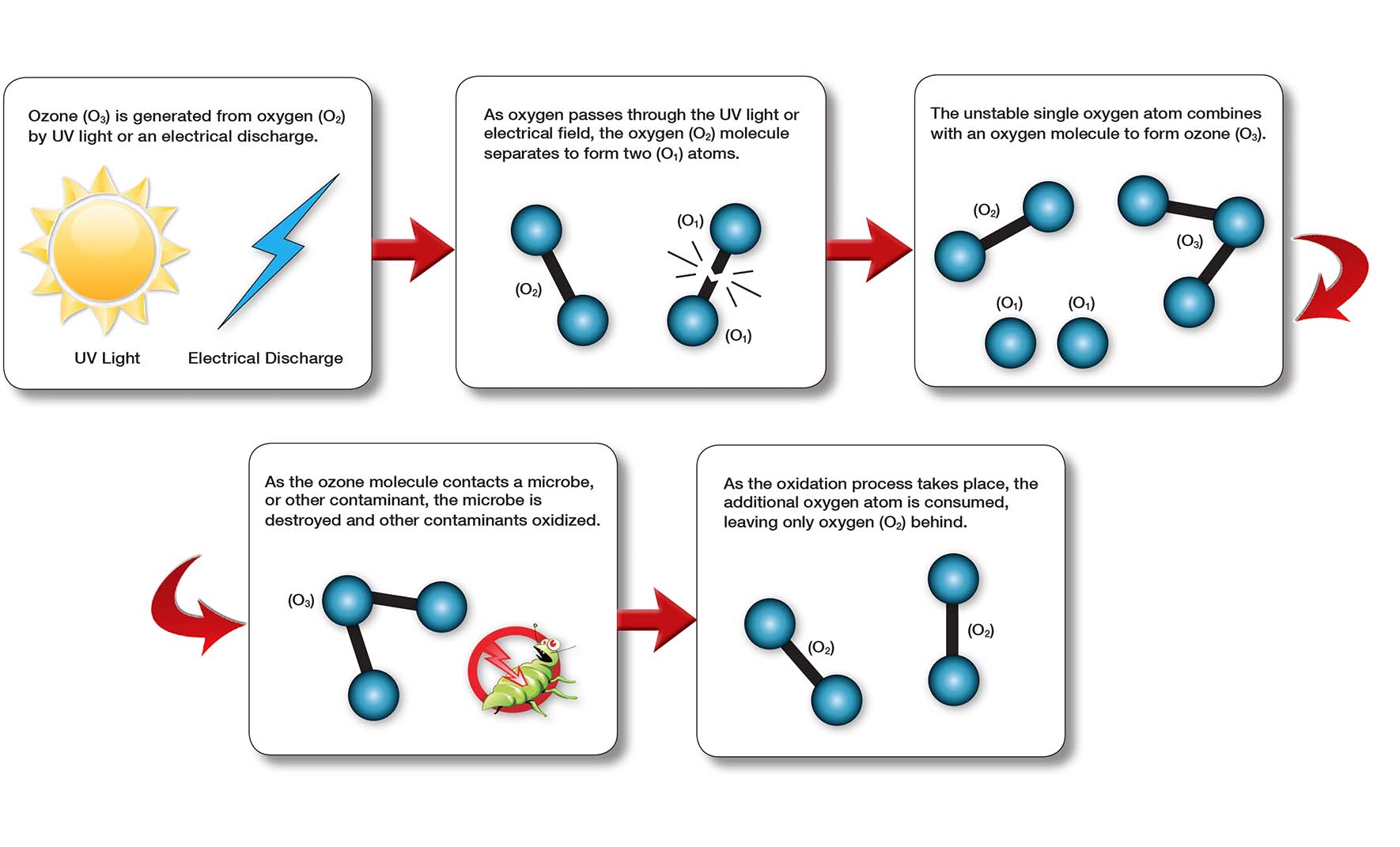
Ozone exists when oxygen (O2) is exposed to ultraviolet light, or exposed to high voltages of lightning. That fresh, clean smell that we notice after a rain storm, is ozone. It provides a natural, clean disinfectant that can also be used in a controlled environment used in water and air treatment applications.
As the oxygen molecules (O2) are exposed to these energy fields, they dissociate and split, forming atoms (O1). These wandering oxygen atoms then recombine with other (O2) molecules in the air stream, forming ozone (O3). Ozone is nothing more than another molecular form of oxygen.
Because ozone is highly reactive, it readily oxidizes (breaks down) organic matter. When ozone encounters another compound, one oxygen atom will break away, attach itself to the compound, and oxidize (clean or purify) it.
Good Ozone vs Bad Ozone
Good ozone protects us from harmful UV radiation from the sun. Another form of ozone we are familiar with is a component of air pollution found in the lower atmosphere. We call that bad ozone. Bad ozone is formed when carbon dioxide given off by cars and industrial fumes reacts with sunlight (UV) at ground level.
Let’s break it down…the formation of bad ozone is created by nitrogen oxides (NOx), methane (CH4), carbon monoxide, and sunlight. About 95 percent of these chemicals come from human activity from the burning of coal, gasoline, and oil in motor vehicles, homes, industries, and power plants. The result is smog, the air pollution we see.
There is a huge misconception that ozone is smog, but it’s not. When smog levels are discussed, ozone is targeted because it is relative to smog conditions and is much easier to measure than other components that smog form. Ozone is actually there to help eliminate smog.
Ozone Benefits
Ozone is an oxidizer and disinfectant commercially available for water and air treatment. It has is approved by the FDA, USDA, and the EPA as antimicrobial disinfectants. Ozone systems require only cold water, reduces the need for chemicals, and are made on-site, on-demand.
With ClearWater Tech’s proven efficiency and reliable ozone systems, you can simplify the handling and storage of chemicals and lower your costs.
It is a more innovative alternative to salt, chlorine, and other chemicals. It can oxidize and reduce microorganisms such as bacteria, fungus, and viruses in water faster than chlorine and faster than bromine. Ozone reduces odor and color. It can also be used for the flocculation of organic material, which simplifies mechanical filtration. It is generated at the point of use and then reverts back to oxygen.
How is Ozone used for Water & Air Purification?
When used correctly, ozone is very effective for many applications, especially at high concentrations. It must be made on-site and used immediately. Ozone generators produce ozone first by compressing ambient air, then separating it and concentrating the oxygen. The oxygen runs through a high-voltage device, called a corona, essentially “lightning in a bottle.” The generator then “injects” the gaseous ozone into the water using negative pressure or a vacuum.
- Disinfection – Reduces bacteria and the inactivation of viruses and cysts.
- Oxidation of Inorganics – Precipitates iron, manganese, sulfides, nitrites, and organically bound heavy metals.
- Oxidation of Organics – Organics causing color, taste, and odor problems, reducing some detergents, phenols, VOCs, turbidity control, and micro flocculation of soluble organics.
There are two Methods to Generate Ozone:
1. Corona Discharge (CD)
2. Ultraviolet (UV) Light
Corona discharge generators are much more sophisticated than UV and can generate much higher levels of ozone. Ozone passes a stream of dry air or oxygen through a high voltage field called a corona discharge, where oxygen in the stream converts to ozone. Ozone generates at the point of application.
UV ozone generators use a light source that generates a narrow band of ultraviolet light. Standard UV ozone generators are less expensive and produce ozone with a concentration of about 0.5% or lower. UV also requires the air (oxygen) to be exposed to the UV source for a more extended amount of time. UV generators are less effective for use in situations that deal with rapidly moving air or water streams.
Ozone Safety
A properly designed and installed ozone system will not expose anyone to irritating ozone concentrations at low levels. However, ozone, like any other potent oxidizing agent, can be harmful if not handled properly.
OSHA – Occupational Safety & Health Administration
OSHA has set specific guidelines for using ozone in the workplace and based on time-weighted averages. Ozone levels should never exceed the following standards: 0.10 ppm (parts per million) for an 8-hour work shift.
• 0.2 ppm for no more than 2 hours exposure
• 0.1 ppm for 8 hours per day exposure doing light work
• 0.08 ppm for 8 hours per day exposure doing moderate work
• 0.05 ppm for 8 hours per day exposure doing heavy work
Ozone Material Safety Data Sheet (MSDS)
The Ozone Material Safety Data Sheet is an official document that includes the properties of ozone; the physical, health, and environmental health hazards; protective measures; and safety precautions for handling, storing, and transporting ozone.
Download our SDS here…
Safety Monitoring Equipment
All ozone monitors are used with ozone generators. They measure the concentration of ozone levels at the permissible exposure limit according to OSHA guidelines. They can shut down the ozone output immediately if necessary.
1. ClearWater Tech’s Ozone Monitor
2. Aeroqual Ambient Ozone Monitor
4. Ozone Safety Badges are a low-cost method that can detect the presence of ozone by forming a color change in the form of an “exclamation mark” which warns the presence of ozone.
5. Ozone Respirators can be a simple paper mask or a gas mask that offers complete protection against any potential adverse effect of overexposure to ozone.